|
What’s the Matter? Elements, Compounds and Mixtures
Anything that has mass and takes up space is matter. Matter is everywhere. Everything you touch is matter. During the time of Aristotle, people thought everything was made up of a combination of air, fire, water and earth. Today, we know that there are a certain number of elements that make up all matter on Earth. Elements are made of tiny particles called atoms. Atoms are composed of a certain number of protons, electrons, and neutrons. Matter has physical and chemical properties. Color, smell, mass, volume, density, temperature, freezing point and boiling point are examples of physical properties of matter. The way elements combine and react with each other are chemical properties. Matter can change physically and chemically. To understand how matter changes, you need to know something about molecules. |
Elements, Compounds, and Mixtures
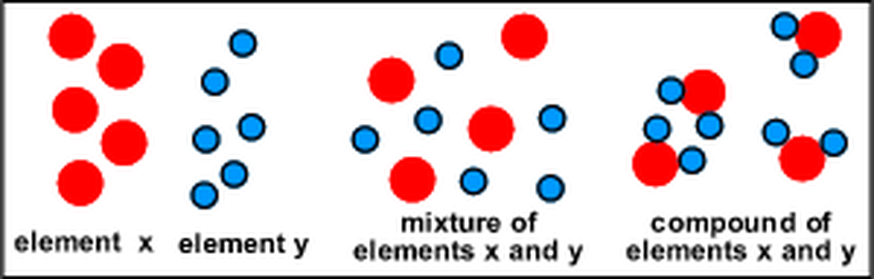
Elements are the building blocks of all matter. They are substances that cannot be broken down or divided by ordinary chemical means. The smallest possible amount of an element is called an atom. We know of over 100 elements. About 92 occur in nature and the rest are man-made. Four of the elements make up 96% of all living matter: carbon, oxygen, hydrogen, and nitrogen. Eight elements make up 99% of the Earth’s crust.
One or two letters represent each element. The elements are arranged in order based on their number of protons in what we call the Periodic Table. They are grouped by their properties. The rows are called periods. Columns are called groups. All elements in the groups have similar chemical and physical properties.
Compounds are substances made of two or more elements that are combined chemically. Compounds can be in the form of a solid, liquid or a gas. They can change from one phase to another, but the elements that combine to make them cannot be broken down through a physical process. For example, when two atoms of hydrogen chemically combine with one atom of oxygen, they form water. The symbol for water, H2O, shows how the atoms combined. Through a physical change, liquid water can lose heat energy and become a solid, or it can gain heat energy and become a gas. Solid water can gain energy and change to the liquid phase. Physical changes do not change the nature of the molecule. But a chemical process can break the water molecule back into hydrogen and oxygen molecules. When atoms combine or molecules are chemically separated, the new substance is very different.
Minerals are other example of compounds. Pyrite, or Fool’s Gold, is a compound that is made of the elements iron and sulfur. Each molecule of Pyrite is made of 1 part of iron and 2 parts of sulfur that are chemically combined. The chemical symbol for Pyrite is FeS2. The mineral magnetite (Fe3O4) is a natural magnet formed when 3 atoms of iron combine chemically with 4 atoms of oxygen. In forming a specific compound, the number and kind of elements always combine in the same ratio or proportion.Chemists look for new ways to chemically combine the elements to make new compounds with properties that they find useful, but elements always remain the same.
Mixtures are a physical combination of two or more elements or compounds. These elements or compounds are not chemically combined. The amount of the substance that combines does not always have to be exactly the same. There is not a set ratio for how they are mixed. For example, you can mix a different amount of sugar with water and still have sugar water. A new substance is not formed in a mixture. The original materials are still there and they can be easily separated by physical means. The properties of the mixture could be similar to the properties of the substances that came together to form them.
There are many kinds of mixtures. Solutions are mixtures in which the particles are spread out evenly throughout the mixture. The particles are very small and will not settle out. Solutions can be made from all phases of matter. Examples of solutions are KoolAid, tea, sugar water and salt water. Mixtures can also be in a solid form. Rocks are an example of a mixture of solids. Minerals combine physically to make rocks, but elements combine chemically to make minerals.
Elements are the building blocks of all matter. They are substances that cannot be broken down or divided by ordinary chemical means. The smallest possible amount of an element is called an atom. We know of over 100 elements. About 92 occur in nature and the rest are man-made. Four of the elements make up 96% of all living matter: carbon, oxygen, hydrogen, and nitrogen. Eight elements make up 99% of the Earth’s crust.
One or two letters represent each element. The elements are arranged in order based on their number of protons in what we call the Periodic Table. They are grouped by their properties. The rows are called periods. Columns are called groups. All elements in the groups have similar chemical and physical properties.
Compounds are substances made of two or more elements that are combined chemically. Compounds can be in the form of a solid, liquid or a gas. They can change from one phase to another, but the elements that combine to make them cannot be broken down through a physical process. For example, when two atoms of hydrogen chemically combine with one atom of oxygen, they form water. The symbol for water, H2O, shows how the atoms combined. Through a physical change, liquid water can lose heat energy and become a solid, or it can gain heat energy and become a gas. Solid water can gain energy and change to the liquid phase. Physical changes do not change the nature of the molecule. But a chemical process can break the water molecule back into hydrogen and oxygen molecules. When atoms combine or molecules are chemically separated, the new substance is very different.
Minerals are other example of compounds. Pyrite, or Fool’s Gold, is a compound that is made of the elements iron and sulfur. Each molecule of Pyrite is made of 1 part of iron and 2 parts of sulfur that are chemically combined. The chemical symbol for Pyrite is FeS2. The mineral magnetite (Fe3O4) is a natural magnet formed when 3 atoms of iron combine chemically with 4 atoms of oxygen. In forming a specific compound, the number and kind of elements always combine in the same ratio or proportion.Chemists look for new ways to chemically combine the elements to make new compounds with properties that they find useful, but elements always remain the same.
Mixtures are a physical combination of two or more elements or compounds. These elements or compounds are not chemically combined. The amount of the substance that combines does not always have to be exactly the same. There is not a set ratio for how they are mixed. For example, you can mix a different amount of sugar with water and still have sugar water. A new substance is not formed in a mixture. The original materials are still there and they can be easily separated by physical means. The properties of the mixture could be similar to the properties of the substances that came together to form them.
There are many kinds of mixtures. Solutions are mixtures in which the particles are spread out evenly throughout the mixture. The particles are very small and will not settle out. Solutions can be made from all phases of matter. Examples of solutions are KoolAid, tea, sugar water and salt water. Mixtures can also be in a solid form. Rocks are an example of a mixture of solids. Minerals combine physically to make rocks, but elements combine chemically to make minerals.
Element Percentage in Earth’s Crust
Oxygen 47%
Silicon 28%
Aluminum 8%
Iron 5%
Calcium 3.5%
Sodium 3%
Potassium 2.5%
Magnesium 2%
All Other Elements Found in the Earth's Crust 1%
Oxygen 47%
Silicon 28%
Aluminum 8%
Iron 5%
Calcium 3.5%
Sodium 3%
Potassium 2.5%
Magnesium 2%
All Other Elements Found in the Earth's Crust 1%
States of Matter
Most matter on Earth exists in one of three states or phases: solid, liquid, or gas.
Each of these states is also known as a phase. The phases differ in how their molecules move. Molecules move randomly and collide with each other in all three phases. In a solid, they are held together in a set pattern by a bond. In a liquid, the bond is not as strong and the molecules can slide past each other. They still stay close to each other and this explains why liquid has a well-defined volume. Since molecules slide past each other in a liquid, they take the shape of the container they are in. There are no bonds that hold the molecules together in a gas. They move independently, filling the space of whatever they are in. Matter moves from one phase to another through a physical process. The state of matter changes when energy is added or taken away.
Most matter on Earth exists in one of three states or phases: solid, liquid, or gas.
Each of these states is also known as a phase. The phases differ in how their molecules move. Molecules move randomly and collide with each other in all three phases. In a solid, they are held together in a set pattern by a bond. In a liquid, the bond is not as strong and the molecules can slide past each other. They still stay close to each other and this explains why liquid has a well-defined volume. Since molecules slide past each other in a liquid, they take the shape of the container they are in. There are no bonds that hold the molecules together in a gas. They move independently, filling the space of whatever they are in. Matter moves from one phase to another through a physical process. The state of matter changes when energy is added or taken away.